Electrodeionization (EDI) is an electrically-driven water treatment technology that uses electricity, ion exchange and resin to remove ionized species from water. The combination of ion-exchange resins and ion-exchange membranes, which are used to move ionic impurities into a waste or concentrate water stream leaving purified product water.

As impurities leave via the concentrate water system, their build-up does not exhaust the resin and therefore prolongs resin lifespan. A single EDI unit may operate for many years before a replacement is required. Typically product water resistivity of >15 MΩ.cm is consistently achieved using this process. This technology can be used as an alternative to single-use purification cartridges.
Its development and use in water purification overcame some of the limitations of ion exchange resin beds, particularly the release of ions as the beds exhaust.
How does it work?
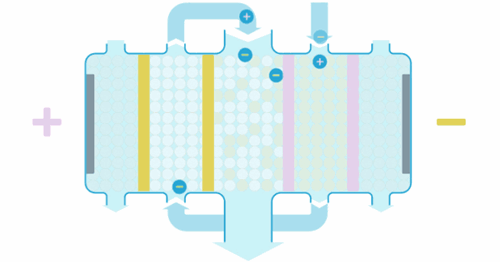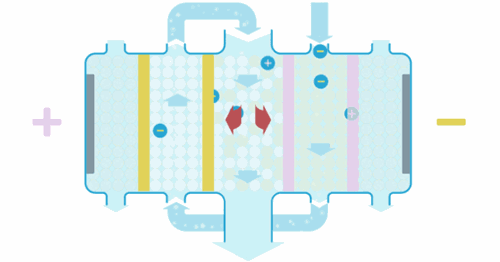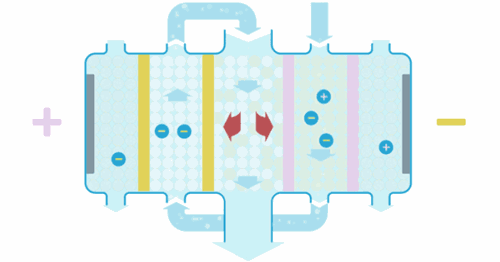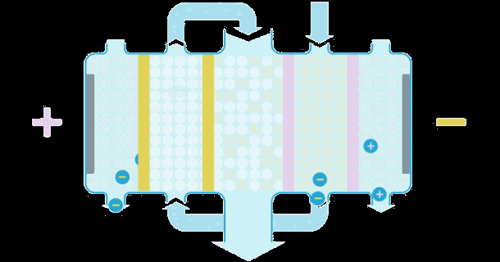
Water enters the EDI module, where an applied current forces ions to move through the resins and across the membranes. These ions are collected into concentrate streams which can then be put to drain or be recycled. The deionized product water can then be used directly or undergo further treatment for enhanced water purity.
When the ions are moved through the resins and between the cation or anion selective membranes, they are exchanged for H+ and OH– ions. Ions that become bound to the ion exchange resins migrate to a separate chamber under the influence of an externally applied electric field. This also produces the H+ and OH– ions necessary to maintain the resins in their regenerated state. Ions in the separate chamber are flushed to waste.

The ion exchange beds in our EDI systems are regenerated continuously so that they do not exhaust in the same way as ion exchange beds that are operated in batch mode.
